Prevention-First
Medicine Starts Here
Pioneering Gut Barrier Protection
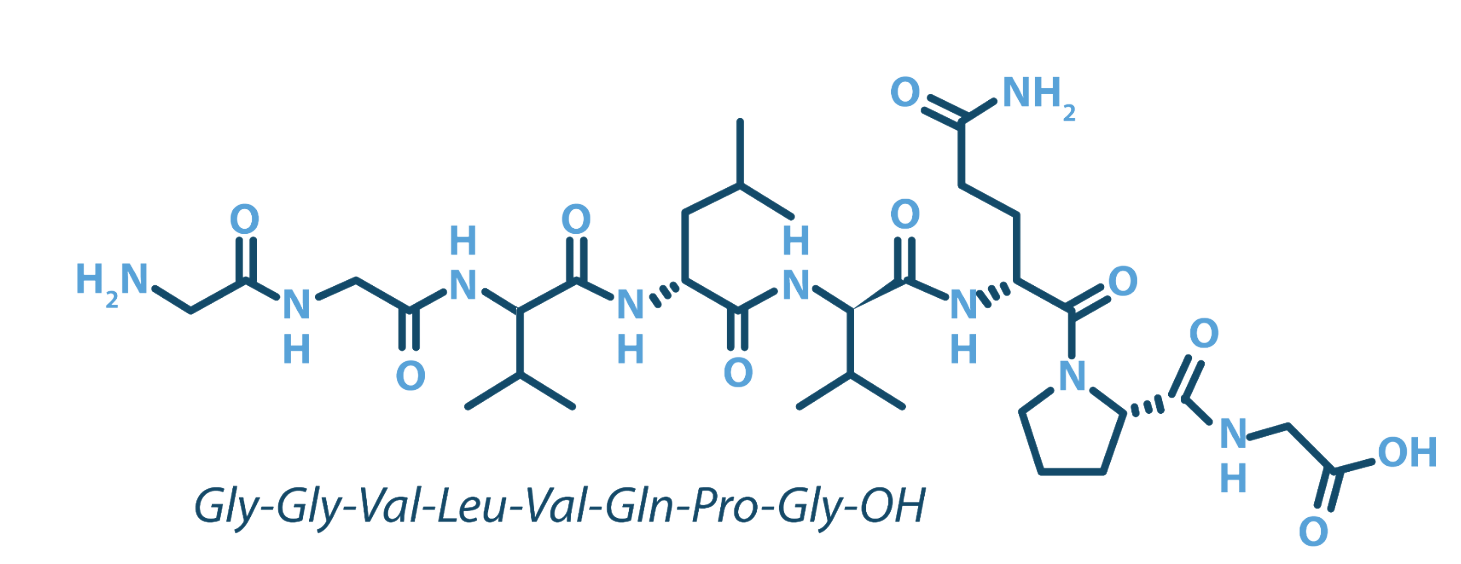
Interlude Biopharma is pioneering therapies that preserve gut integrity and prevent inflammation at its source. Our lead candidate, Larazotide Acetate, is a Phase 3 ready, first ever therapy designed to protect patients with celiac disease from gluten-induced immune activation — a breakthrough for millions living without an approved treatment today.
Leading the Evolution of Tight Junction Science
Interlude is advancing first-in-class, non-immunosuppressive therapy designed to safeguard mucosal and epithelial integrity. Our work centers on tight-junction regulation—a pioneering approach that preserves gut wall integrity and prevents inflammation at its source, rather than just treating the symptoms.
Our Pipeline: One Mechanism. Many Diseases. Limitless Potential
Our pipeline extends prevention‑first science to a broad spectrum of diseases driven by epithelial disruption. By stabilizing the body's natural barriers, we are addressing the root cause of autoimmune, inflammatory, and post‑infectious conditions.
Prevention at the Source
Chronic inflammation begins when intestinal barriers fail. Larazotide is the first therapy designed to keep them intact.
The Problem: Barrier Breakdown
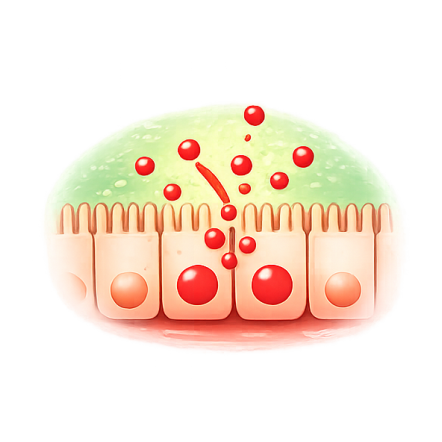
Your intestinal lining uses protein "gatekeepers" called tight junctions to control what passes from the gut into the bloodstream. In celiac disease, gluten triggers zonulin release, which opens these tight junctions.
Once open, gluten fragments cross into tissues, triggering immune cells that cause inflammation, villous atrophy, and the debilitating symptoms patients experience: abdominal pain, diarrhea, fatigue, malabsorption, and increased risk of complications.
The Solution: Upstream Intervention

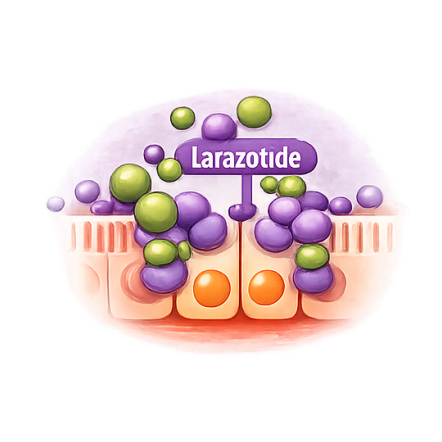
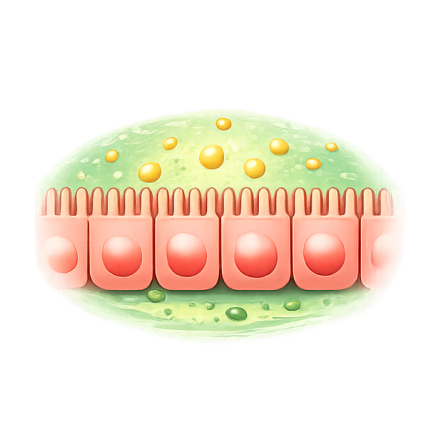
Larazotide Acetate Is a First-Ever Prevention Therapy for Celiac Disease — and Beyond.
A Late-Stage Opportunity in Celiac Disease
Mechanism
Competitively inhibits zonulin receptor, preventing tight junction opening and maintaining intestinal barrier integrity
Formulation
Oral peptide acts locally in the gut where it’s needed most.
Patient Benefit
Protection against inadvertent gluten exposure, enabling dietary flexibility and improved quality of life
Freedom to Eat. Confidence to Live.
People with celiac disease live in constant vigilance — every meal a potential risk. Cross-contamination can mean days of pain, fatigue, and anxiety.
Living with Celiac Disease
Larazotide Acetate is an investigational therapy designed to help strengthen the gut's natural defense, with the goal of reducing the immune activation that can begin when gluten exposure occurs — giving patients the confidence to share meals, travel, and reconnect socially. Protecting gut integrity means protecting every meal, every day.
Participating in Our Research
Interlude Biopharma is committed to continue to advance innovative treatments for
patients living with celiac disease and related conditions. We are actively conducting
clinical trials to evaluate our investigational therapies.
Phase 3 Ready. FDA Fast Track.
Why Now?
Celiac disease represents one of the largest untreated chronic conditions in gastroenterology. Despite affecting 1 in 70 Americans, zero FDA-approved therapies exist—leaving patients with diet alone.
Diagnosed Celiac Patients (US)
1.4% prevalence rate, growing annually as awareness and diagnosis rates improve. Current standard of care: gluten-free diet alone.
Remain Symptomatic on GFD
Gluten-free diet is insufficient for majority. Cross-contamination is common despite best efforts.
Peak Sales Potential
Based on 30% penetration of diagnosed celiac market.